- Access to PreHevbrio [Hepatitis B Vaccine (Recombinant)] continues to broaden in the U.S. and beyond, with additional approvals received in 2022 in Europe and Canada
- Clinical data expected throughout 2023 from: (i) VBI-2601: two ongoing Phase II studies in chronically infected hepatitis B (HBV) patients, and (ii) VBI-2901: first clinical study of multivalent vaccine candidate designed to increase breadth of protection against COVID-19 and related coronaviruses
- Additional clinical studies expected to initiate in Q2 and mid-year 2023 assessing VBI-1901 in both the recurrent and primary glioblastoma (GBM) settings, respectively
VBI Vaccines Inc. (Nasdaq: VBIV) (VBI), a biopharmaceutical company driven by immunology in the pursuit of powerful prevention and treatment of disease, today provided a business update and announced financial results for the year ended December 31, 2022.
Jeff Baxter, VBI’s President and CEO commented: “The achievements of 2022, across our pipeline, set 2023 up for a strong start, and I am encouraged by the building momentum for PreHevbrio as well as for our development-stage candidates targeting chronic hepatitis B, glioblastoma, and broader protection against COVID-19. We remain intensely focused on three priorities: (i) making a difference in the fight against hepatitis B, (ii) advancing our pipeline candidates targeting additional significant medical and public health needs, and (iii) managing our operating expenses and capital to fuel sustainable growth and value for key stakeholders – patients, healthcare providers, and shareholders. The potential impact our programs can have on patients and public health grows each year, and I am excited for the continued evolution of VBI in 2023.”
Notable Recent Program Achievements and Projected Upcoming Milestones
Hepatitis B (HBV): Working to Address Both Sides of the Fight Against HBV – Prevention and Treatment
PreHevbrio™ [Hepatitis B Vaccine (Recombinant)]
- Net product sales of $0.9 million in 2022, following the U.S. launch at the end of Q1 2022
- In Q1 2023, product access agreements were put in place with six retail pharmacy chains, including three of the top 10 national retail chains and three of the top 10 regional retail pharmacy networks
- Reimbursement coverage is now estimated to be in place for 75% of Medicare-insured lives, 75% of commercially insured lives, and more than 60% of lives under state Medicaid plans
- Momentum for PreHevbrio continues to build, with customer acquisition rate more than doubling since the beginning of Q4 2022
Outside of the U.S.:
- Now approved in the United Kingdom, European Union/European Economic Area (brand name PreHevbri®), and in Canada (brand name PreHevbrio)
- H1 2023: As part of our partnership with Valneva, announced in September 2022, we expect PreHevbri will be available in certain European markets beginning in the first half of 2023
- 2023: We expect to make PreHevbrio available in Canada beginning in 2023
VBI-2601 (BRII-179): HBV Immunotherapeutic Candidate
- Initial Phase 2 data, announced in February 2023, suggest VBI-2601 in combination with BRII-835, an HBV-targeting siRNA, induced stronger anti-hepatitis B surface antigen (HBsAg)-specific T-cell and antibody responses compared to the siRNA alone; to date, two patients who received the combination regimen achieved maximum reductions in HBsAg at or below the lower limit of quantification (LLOQ) by Week 40
- Represents strong HBV-specific immunomodulator data and suggests that VBI-2601 has the potential to be part of a functional cure regimen
- Later in 2023: Additional data from this combination study expected to be announced
- Q3 2023: Interim topline data expected from part one of the two-part Phase 2a/2b combination study evaluating VBI-2601 (BRII-179) as an add-on to existing pegylated interferon (PEG-IFN-α) and nucleos(t)ide reverse transcriptase inhibitor (Nrtl) therapy in non-cirrhotic chronic HBV patients
Glioblastoma (GBM): Survival Benefit and Tumor Responses Observed – Moving to Next Phase of Development
VBI-1901: Cancer Vaccine Immunotherapeutic Candidate
- Program has now received both U.S. FDA Orphan Drug Designation (announced in June 2022) and FDA Fast Track Designation (announced in June 2021) for the treatment of GBM
- Data from the Phase II study, as presented throughout 2022, demonstrated improvement in 6-, 12-, and 18-month overall survival compared to historical controls, two partial tumor responses (with one patient on protocol for more than two years achieving a maximum tumor reduction of 93%), and 10 stable disease observations
- Based on the data seen to-date, we anticipate assessing VBI-1901 in randomized, controlled studies in both the primary and recurrent GBM settings
- Q2 2023: Expected initiation of next phase of development in recurrent GBM setting
- Mid-Year 2023: Expected initiation of VBI-1901 study arm, as part of the Individualized Screening Trial of Innovative Glioblastoma Therapy (INSIGhT), a Phase 2 adaptive platform trial, in combination with Agenus’ anti-PD-1, balstilimab, in the primary GBM setting
COVID-19 & Coronaviruses: Targeting Broader Immunity – Clinical Data Expected Mid-Year 2023
VBI-2901: Multivalent Coronavirus Vaccine Candidate
- Partnership with the Coalition for Epidemic Preparedness Innovations (CEPI) expanded to advance the development of multivalent coronavirus vaccines that could be deployed against COVID-19 as well as future “Coronavirus X” (announced in December 2022)
- Mid-year 2023: Interim data expected from Phase 1 study of VBI-2901, VBI’s vaccine candidate that expresses the SARS-CoV-2 (COVID-19), SARS-CoV-1 (SARS), and MERS-CoV (MERS) spike proteins
Additional Corporate Updates
- Debt Financing: In September 2022, VBI closed a refinanced and upsized debt facility of up to $100 million with existing lender, K2 HealthVentures (K2HV), adding $20 million of non-dilutive funding to the balance sheet
Financial Results for the Twelve Months Ended December 2022
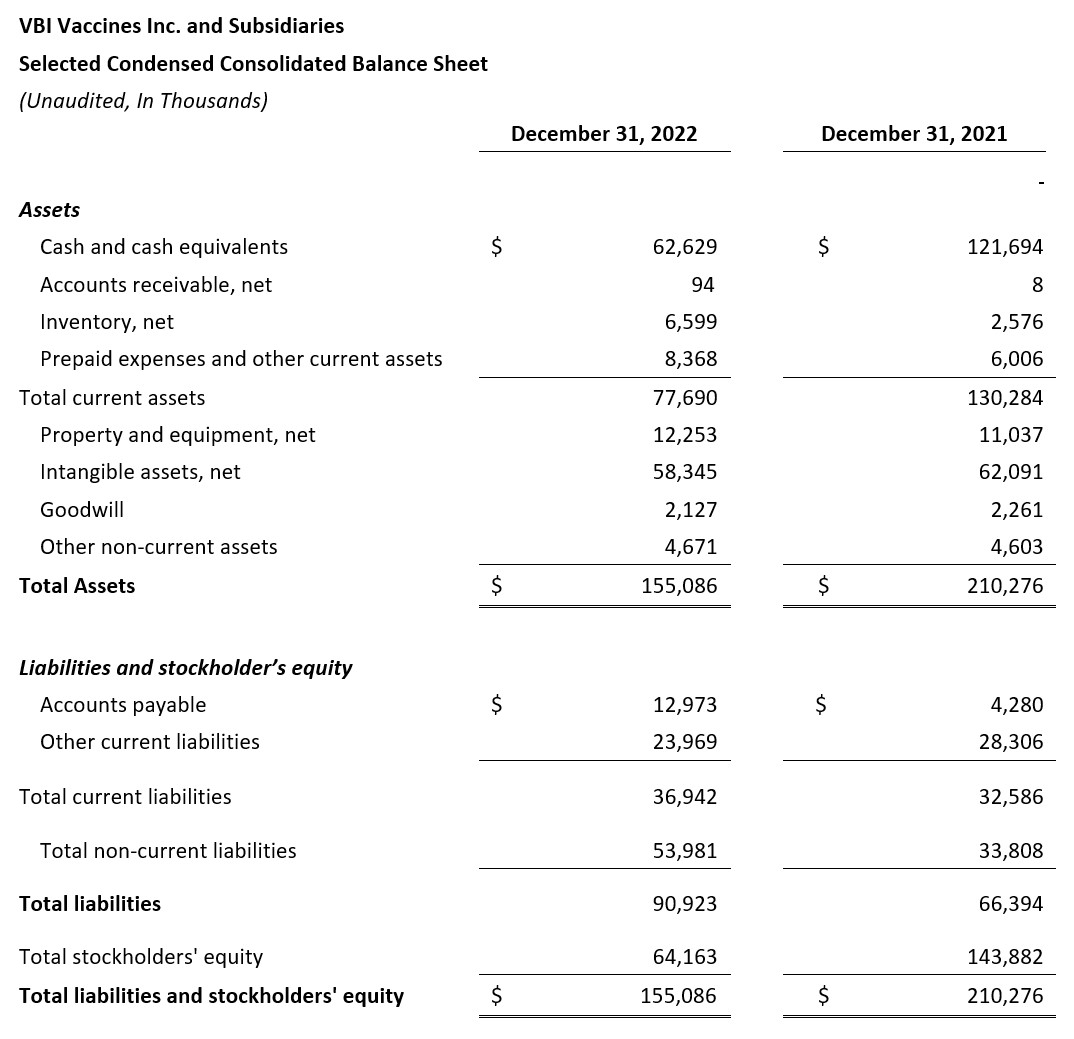
- Cash Position: As of December 31, 2022 VBI had $62.6 million in cash compared with $121.7 million in cash as of December 31, 2021.
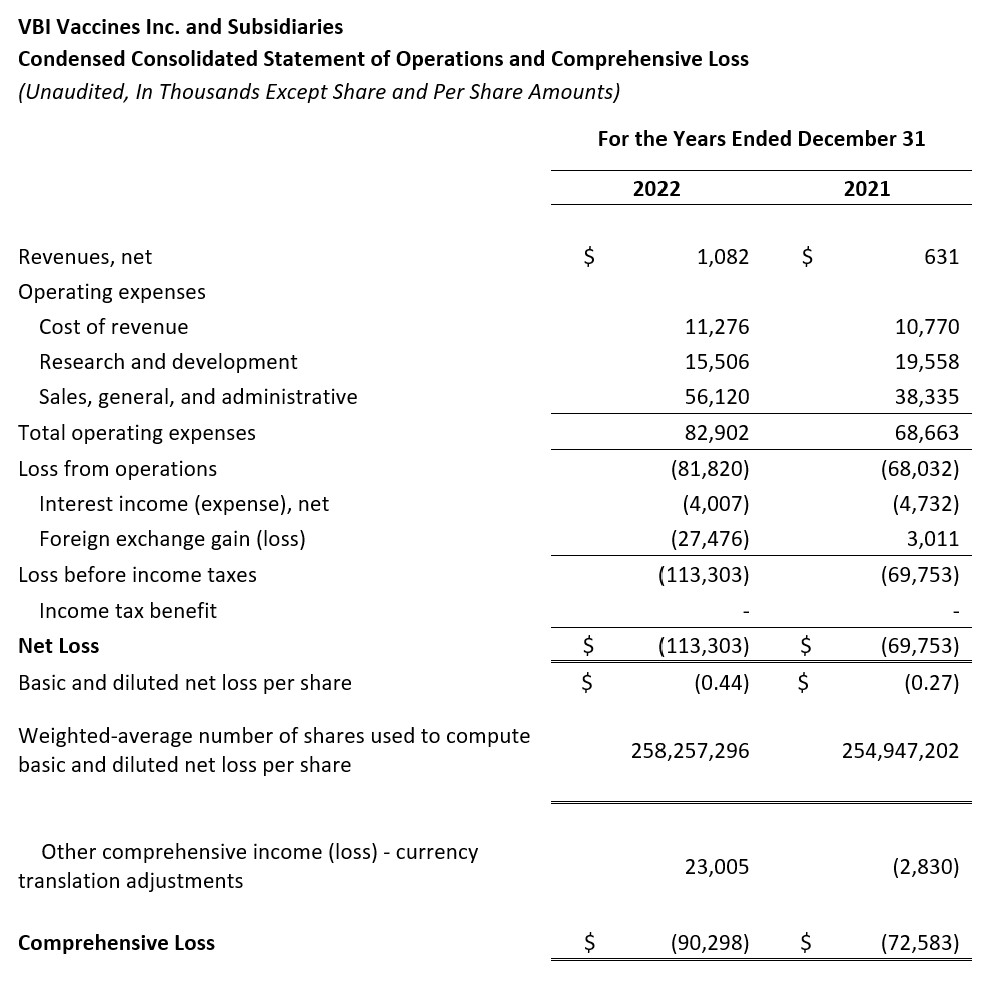
- Revenues, net: Revenues, net for the full year 2022 were $1.1 million, compared to $0.6 million for the same period in 2021. The increase was due to an increase in U.S. product revenue related to the launch of PreHevbrio in the U.S.
- Cost of Revenues: Cost of revenues was $11.3 million for the full year 2022 as compared to $10.8 million in 2021. The increase was due to increased outsourced testing costs, direct labor costs, and increased inventory related costs for our 3-antigen HBV vaccine.
- Research and Development (R&D): R&D expenses for the year ended December 2022 were $15.5 million compared to $19.6 million in 2021. R&D expenses were offset by $8.9 million and $14.9 million in government grants and funding arrangements, in 2022 and 2021, respectively. The decrease in R&D expenses related to a refund of $2.9 million received from the FDA related to the Prescription Drug User Fee Act program fee for PreHevbrio and a decrease in costs for our coronavirus vaccine program, offset by an increase in R&D expenses for VBI-1901 as we prepare for clinical studies in primary and recurrent GBM patients.
- Sales, General, and Administrative (G&A): SG&A expenses for the full year 2022 were $56.1 million compared to $38.3 million for the full year 2021. The increase in SG&A expenses, partially offset by government grants and funding arrangements, was a result of the increased commercial activities related to PreHevbrio in the U.S., most notably the deployment of our U.S. promotional field team and development of our distribution infrastructure. Additional increased costs include increased insurance costs, increased professional costs, and increased labor costs.
- Net Cash Used in Operating Activities: Net cash used in operating activities for the full year 2022 was $73.7 million, compared to $39.9 million for the same period in 2021. The increase in cash outflows was largely due to commercial expenses for the launch of PreHevbrio in the U.S, increased usage of cash as we build inventory for continued commercialization, less funding received from CEPI, and changes in other operating working capital balances. Additionally, there was $1.0 million cash advanced from CEPI pursuant to the CEPI Funding Agreement during 2022 compared to $18.4 million cash advanced from the CEPI Funding Agreement in 2021.
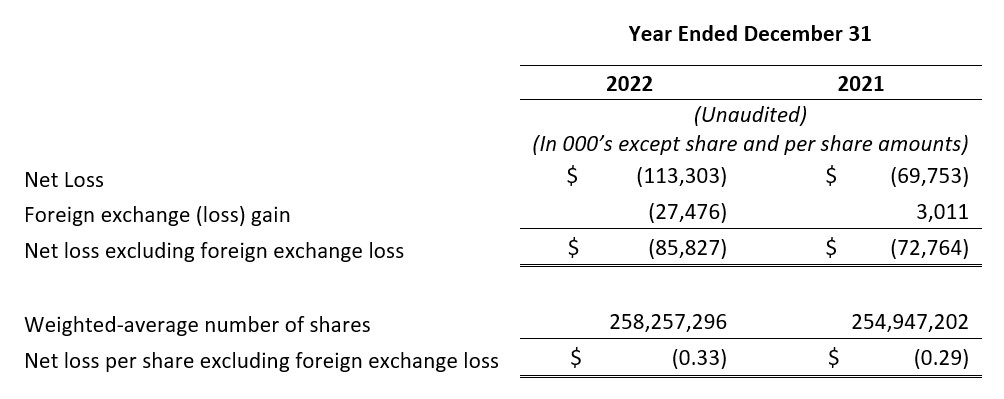
- Net Loss and Net Loss Per Share: Net loss and net loss per share for the full year 2022 were $113.3 million and $0.44, respectively, compared to a net loss of $69.8 million and a net loss per share of $0.27 for the full year 2021.
- Net Loss and Net Loss Per Share, Excluding Foreign Exchange Loss: Net loss and net loss per share, excluding foreign exchange loss, for the full year 2022 were $85.8 million and $0.33, respectively, compared to a net loss and a net loss per share, excluding foreign exchange loss, of $72.8 million and $0.29 for the full year 2021. Foreign exchange loss for the full year 2022 was $27.5 million as compared to a gain of $3.0 million for the full year 2021. Certain intercompany loans between VBI Vaccines Inc. and our subsidiaries are denominated in a currency other than the functional currency of each entity. The primary driver of the increase in foreign exchange loss was the impact of the relative strengthening of the U.S. and Canadian Dollars against the New Israeli Shekel upon translation of these intercompany loans.
Use of Non-GAAP Financial Measures
Net Loss Excluding Foreign Exchange Loss and Net Loss per Share Excluding Foreign Exchange Loss are non-GAAP financial measures. VBI’s management believes that the presentation of Net Loss Excluding Foreign Exchange Loss and Net Loss per Share Excluding Foreign Exchange Loss is useful to investors because management does not consider foreign exchange loss, which is primarily driven by changes in exchange rates related to certain intercompany loans, when evaluating VBI’s operating performance. Non-GAAP financial measures are meant to supplement, and to be viewed in conjunction with, GAAP financial results. The presentation of these non-GAAP financial measures should not be considered in isolation or as a substitute for comparable GAAP financial measures and should be read only in conjunction with the Company’s financial statements prepared in accordance with GAAP. Reconciliations of the Company’s non-GAAP measures are included below.
The following represents a reconciliation of Net Loss to Net Loss Excluding Foreign Exchange Loss and Net Loss per Share Excluding Foreign Exchange Loss.
About PreHevbrio [Hepatitis B Vaccine (Recombinant)]
PreHevbrio is the only 3-antigen hepatitis B vaccine, comprised of the three surface antigens of the hepatitis B virus – S, pre-S1, and pre-S2. It is approved for use in the U.S., European Union/European Economic Area, United Kingdom, Canada, and Israel. The brand names for this vaccine are: PreHevbrio™ (US/Canada), PreHevbri® (EU/EEA/UK), and Sci-B-Vac® (Israel).
Please visit www.PreHevbrio.com for U.S. Important Safety Information for PreHevbrio [Hepatitis B Vaccine (Recombinant)], or please see U.S. Full Prescribing Information.
U.S. Indication
PreHevbrio is indicated for prevention of infection caused by all known subtypes of hepatitis B virus. PreHevbrio is approved for use in adults 18 years of age and older.
U.S. Important Safety Information (ISI)
Do not administer PreHevbrio to individuals with a history of severe allergic reaction (e.g. anaphylaxis) after a previous dose of any hepatitis B vaccine or to any component of PreHevbrio.
Appropriate medical treatment and supervision must be available to manage possible anaphylactic reactions following administration of PreHevbrio.
Immunocompromised persons, including those on immunosuppressant therapy, may have a diminished immune response to PreHevbrio.
PreHevbrio may not prevent hepatitis B infection, which has a long incubation period, in individuals who have an unrecognized hepatitis B infection at the time of vaccine administration.
The most common side effects (> 10%) in adults age 18-44, adults age 45-64, and adults age 65+ were pain and tenderness at the injection site, myalgia, fatigue, and headache.
There is a pregnancy exposure registry that monitors pregnancy outcomes in women who received PreHevbrio during pregnancy. Women who receive PreHevbrio during pregnancy are encouraged to contact 1-888-421-8808 (toll-free).
To report SUSPECTED ADVERSE REACTIONS, contact VBI Vaccines at 1-888-421-8808 (toll-free) or VAERS at 1-800-822-7967 or www.vaers.hhs.gov.
Please see Full Prescribing Information.
About VBI Vaccines Inc.
VBI Vaccines Inc. (“VBI”) is a biopharmaceutical company driven by immunology in the pursuit of powerful prevention and treatment of disease. Through its innovative approach to virus-like particles (“VLPs”), including a proprietary enveloped VLP (“eVLP”) platform technology, VBI develops vaccine candidates that mimic the natural presentation of viruses, designed to elicit the innate power of the human immune system. VBI is committed to targeting and overcoming significant infectious diseases, including hepatitis B, coronaviruses, and cytomegalovirus (CMV), as well as aggressive cancers including glioblastoma (GBM). VBI is headquartered in Cambridge, Massachusetts, with research operations in Ottawa, Canada, and a research and manufacturing site in Rehovot, Israel.
VBI Contact
Nicole Anderson
Director, Corporate Communications & IR
Phone: (617) 830-3031 x124
Email: IR@vbivaccines.com
Cautionary Statement on Forward-looking Information
Certain statements in this press release that are forward-looking and not statements of historical fact are forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and are forward-looking information within the meaning of Canadian securities laws (collectively, “forward-looking statements”). The Company cautions that such forward-looking statements involve risks and uncertainties that may materially affect the Company’s results of operations. Such forward-looking statements are based on the beliefs of management as well as assumptions made by and information currently available to management. Actual results could differ materially from those contemplated by the forward-looking statements as a result of certain factors, including but not limited to, the impact of general economic, industry or political conditions in the United States or internationally; the impact of the COVID-19 pandemic and the continuing effects of the COVID-19 pandemic on our clinical studies, manufacturing, business plan, and the global economy; the ability to successfully manufacture and commercialize PreHevbrio/PreHevbri; the ability to establish that potential products are efficacious or safe in preclinical or clinical trials; the ability to establish or maintain collaborations on the development of pipeline candidates and the commercialization of PreHevbrio/PreHevbri; the ability to obtain appropriate or necessary regulatory approvals to market potential products; the ability to obtain future funding for developmental products and working capital and to obtain such funding on commercially reasonable terms; the Company’s ability to manufacture product candidates on a commercial scale or in collaborations with third parties; changes in the size and nature of competitors; the ability to retain key executives and scientists; and the ability to secure and enforce legal rights related to the Company’s products. A discussion of these and other factors, including risks and uncertainties with respect to the Company, is set forth in the Company’s filings with the SEC and the Canadian securities authorities, including its Annual Report on Form 10-K filed with the SEC on March 13, 2023, and filed with the Canadian security authorities at sedar.com on March 13, 2023, as may be supplemented or amended by the Company’s Quarterly Reports on Form 10-Q. Given these risks, uncertainties and factors, you are cautioned not to place undue reliance on such forward-looking statements, which are qualified in their entirety by this cautionary statement. All such forward-looking statements made herein are based on our current expectations and we undertake no duty or obligation to update or revise any forward-looking statements for any reason, except as required by law.









