- Prophylactic Hepatitis B (HBV):
- FDA approval of PreHevbrio™ [Hepatitis B Vaccine (Recombinant)] received in November 2021
- PreHevbrio added to ACIP list of recommended adult HBV vaccines in February 2022
- U.S. commercial launch of PreHevbrio expected at the end of Q1 2022
- Positive EMA CHMP opinion received in February 2022
- Ex-U.S. regulatory decisions expected throughout 2022
- Therapeutic HBV:
- Following positive data announced in 2021, two Phase 2 studies initiated in April 2021 and January 2022
- Initial data from Phase 2 studies expected in H2 2022 and H1 2023
- COVID-19 & Coronaviruses:
- New clinical data from VBI-2905 (Beta variant) expected around the end of Q1 2022
- First clinical study of VBI-2901 (multivalent, pan-coronavirus candidate) expected to initiate mid-year 2022
- Glioblastoma (GBM):
- Following positive Phase 1/2a study data, FDA granted Fast Track Designation for VBI-1901 in June 2021
- Two randomized, controlled clinical studies to evaluate VBI-1901 in both recurrent and primary GBM settings expected to initiate in Q2 and H2 2022, respectively
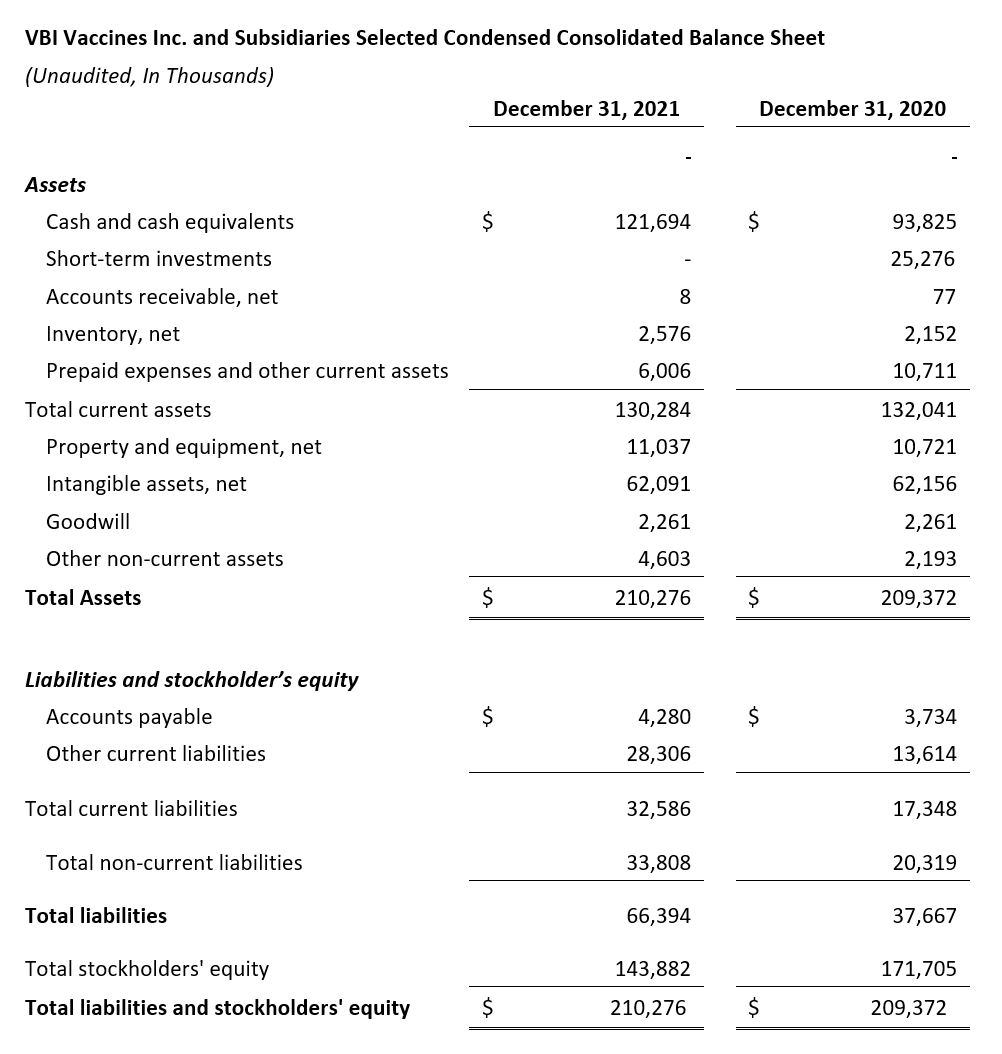
- $121.7 million in cash at year-end 2021
VBI Vaccines Inc. (Nasdaq: VBIV) (VBI), a biopharmaceutical company driven by immunology in the pursuit of powerful prevention and treatment of disease, today announced financial results for the fourth quarter and twelve months ended December 31, 2021. The Company also provided a corporate update and its outlook for 2022.
Annual Note from Jeff Baxter, President and CEO:
“Despite the ongoing impact and disruption of the pandemic, 2021 was a remarkable year for VBI with notable achievements and advancement across all of our lead programs. Our fight against hepatitis B was bolstered by the FDA approval of the only 3-antigen HBV vaccine for adults in the United States, PreHevbrio, and by its subsequent inclusion in the list of ACIP-recommended adult HBV vaccines in February. On the therapeutic HBV front, VBI-2601 progressed into two Phase 2 studies in chronically infected adults, including the first clinical study in the field to evaluate the combination of an HBV vaccine immunotherapeutic candidate and an siRNA candidate. Hepatitis B remains a persistent public health problem and an effective solution will require both prevention and treatment options.
In preparation for the commercial launch of PreHevbrio in the U.S., we have partnered with Syneos Health since 2019. Over the last 18 months, we have expanded this relationship to build the leadership team and now also have more than 80 field-based team members fully dedicated to VBI. The experience, expertise, and drive across the team is a true testament to this partnership and our combined commitment to the fight against HBV. With the commercial team ready to go, we look forward to making PreHevbrio available in the U.S., with an expected commercial launch at the end of March 2022.
COVID-19, as well as the collective response to the pandemic, continues to evolve with ongoing updates to our understanding and knowledge of the current and potential long-term situation. We remain steadfast in our belief that the next-generation of COVID-19 vaccines should not chase variants, but rather add meaningful clinical benefit by broadening immunity to coronaviruses and SARS-CoV-2 variants of concern. The clinical data from the ongoing study of VBI-2905, our monovalent eVLP candidate targeting the Beta variant, is expected to further inform our understanding of the eVLP platform for betacoronaviruses. We are excited to initiate the first clinical study of our multivalent, pan-coronavirus eVLP candidate, VBI-2901, expected mid-year.
Glioblastoma remains one of the most devastating malignant tumors with very few treatment options available to patients. We continue to be encouraged by the data we have seen to-date in clinical studies of VBI-1901, our GBM immunotherapeutic candidate, with two partial tumor responses, one of which remains on protocol past week 100 with a 93% tumor reduction compared to baseline measured at the beginning of treatment, and seven stable disease observations. On the strength of these data, and the substantial unmet need for effective treatment options, the FDA granted VBI-1901 Fast Track Designation in the recurrent setting in June 2021. We are driving hard to initiate the next stages of clinical development, which are expected to be two randomized, controlled studies in the recurrent and the primary GBM settings, increasing our opportunity to help more patients at each step of the GBM diagnosis.
We are committed to being part of the fight against these significant unmet medical and public health needs and the achievements of 2021 have further strengthened our promise. We have already continued this momentum into the early part of 2022, and we look forward to another transformational year ahead.”
Recent Key Program Achievements and Projected Upcoming Milestones
Hepatitis B (HBV)
3-Antigen HBV Vaccine
- November 2021: S. Food and Drug Administration (FDA) approved PreHevbrio [Hepatitis B Vaccine (Recombinant)] for the prevention of infection caused by all known subtypes of HBV in adults age 18 years and older
- December 2021: New Drug Submission (NDS) for the 3-Antigen HBV vaccine filed with Health Canada; marketing authorization decision expected in 2022
- February 2022: PreHevbrio recommended by CDC Advisory Committee on Immunization Practices (ACIP), joining the list of recommended products for prophylactic adult vaccination against HBV infection
- February 2022: Positive opinion received by European Medicines Agency (EMA) Committee for Medicinal Products for Human Use (CHMP) under brand name PreHevbri™ [Hepatitis B vaccine (recombinant, adsorbed)], with final decision on marketing authorization expected from the European Commission (EC) in the coming months
- The United Kingdom Medicines and Healthcare products Regulatory Agency (MHRA) review, through the EC Decision Reliance Procedure (ECDRP), was initiated upon receipt of positive CHMP opinion
- End of Q1 2022: PreHevbrio S. product availability and commercial launch expected, through partnership with Syneos Health, with revenue generation expected to begin in Q2
VBI-2601 (BRII-179): HBV Immunotherapeutic Candidate
- Throughout 2021: Data announced from completed Phase 1b/2a study that demonstrated the ability of VBI-2601 (BRII-179) to induce both B cell (antibody) and T cell responses in chronically infected HBV patients, with a well-tolerated safety profile
- April 2021: Phase 2 combination study initiated to evaluate safety and efficacy of VBI-2601 (BRII-179) in combination with BRII-835 (VIR-2218), an HBV-targeting small interfering ribonucleic acid (siRNA)
- H2 2022: Initial data from Phase 2 combination study expected
- January 2022: Initiation of a second, two-part Phase 2a/2b study to assess safety and efficacy of VBI-2601 (BRII-179) as an “add-on” therapy to standard-of-care treatment in chronic HBV patients – standard-of-care treatment currently has a functional cure rate of approximately 9%1
- H1 2023: Initial data from the Phase 2a/2b “add-on” study expected
COVID-19 & Coronaviruses
VBI-2900: eVLP Coronavirus Vaccine Program
- March 2021: Initiation of Phase 1/2 study of VBI-2902 (monovalent eVLP, ancestral strain)
- March 2021: Announcement of partnership with the Coalition for Epidemic Preparedness Innovations (CEPI), with support from CEPI of up to $33 million to support development of VBI’s eVLP vaccine candidates against SARS-CoV-2 variants
- June 2021: Announcement of positive data from Phase 1a portion of ongoing study of VBI-2902, demonstrating induction of neutralization titers in 100% of participants, with 4.3x higher geometric mean titer (“GMT”) than the convalescent serum panel (n=25), and peak antibody binding GMT of 1:4047
- September 2021: Phase 1b portion of ongoing study initiated with VBI-2905 (monovalent eVLP, Beta variant) as both a 1-dose booster in individuals previously immunized with an mRNA vaccine and as a primary 2-dose series in unvaccinated adults
- Around the end of Q1 2022: Initial data from Phase 1b portion of study with VBI-2905 as a 1-dose booster expected, dependent upon receipt of data from third party clinical research organizations
- Mid-year 2022: Expected initiation of first clinical study of multivalent, pan-coronavirus candidate, VBI-2901
Glioblastoma (GBM)
VBI-1901: Cancer Vaccine Immunotherapeutic Candidate
- Throughout 2021: Data from ongoing Phase 1/2a study announced, demonstrating improvement in 6-month and 12-month overall survival (“OS”) data compared to historical controls and meaningful tumor response data
- June 2021: Based on data from the Phase 1/2a study, FDA granted Fast Track Designation for VBI-1901 formulated with GM-CSF for the treatment of recurrent GBM patients with first tumor recurrence
- Q2 2022: Expected initiation of next phase of development in recurrent GBM setting, aiming to expand the number of patients in the ongoing Phase 1/2a study and adding a control arm, with the potential for accelerated approval based on tumor response rates and improvement in overall survival
- H2 2022: Evaluation of VBI-1901 in the primary GBM setting expected to initiate as part of the Individualized Screening Trial of Innovative Glioblastoma Therapy (INSIGhT), a Phase 2 adaptive platform trial – data from which have potential to support an accelerated approval application
Other Corporate Updates
Financing
- During the year ended December 31, 2021, VBI raised total gross proceeds of $33.3 million, issuing 9.1 million shares at an average price of $3.64 through its Open Market Sales AgreementSM, established July 31, 2020 with Jefferies LLC
- May 2021: Announcement of agreement to draw down a $12 million second tranche under the previously announced debt financing facility with K2 HealthVentures (K2HV), a healthcare-focused specialty finance company
Publications
- May 2021: The Lancet Infectious Diseases publication of data from PROTECT – link here
- June 2021: Vaccine publication of data from Phase 3 study of 3-antigen HBV vaccine in Vietnam – link here
- July 2021: Vaccine publication of data from COVID-19 preclinical studies of VBI-2902 – link here
- September 2021: JHEP Reports publication of Tx HBV data from Phase 1a/2b study of VBI-2601 – link here
- October 2021: JAMA Network Open publication of data from CONSTANT – link here
Financial Results for the Three and Twelve Months Ended December 2021
- Cash Position: VBI ended the fourth quarter of 2021 with $121.7 million in cash compared with $119.1 million in cash, cash equivalents, and short-term investments as of December 31, 2020.
- Net Cash Used in Operating Activities: Net cash used in operating activities for the full year 2021 was $39.9 million, compared to $47.1 million for the same period in 2020.
- Cash Used for Purchase of Property and Equipment: The purchase of property and equipment in 2021 was $2.0 million compared to $1.0 million in 2020.
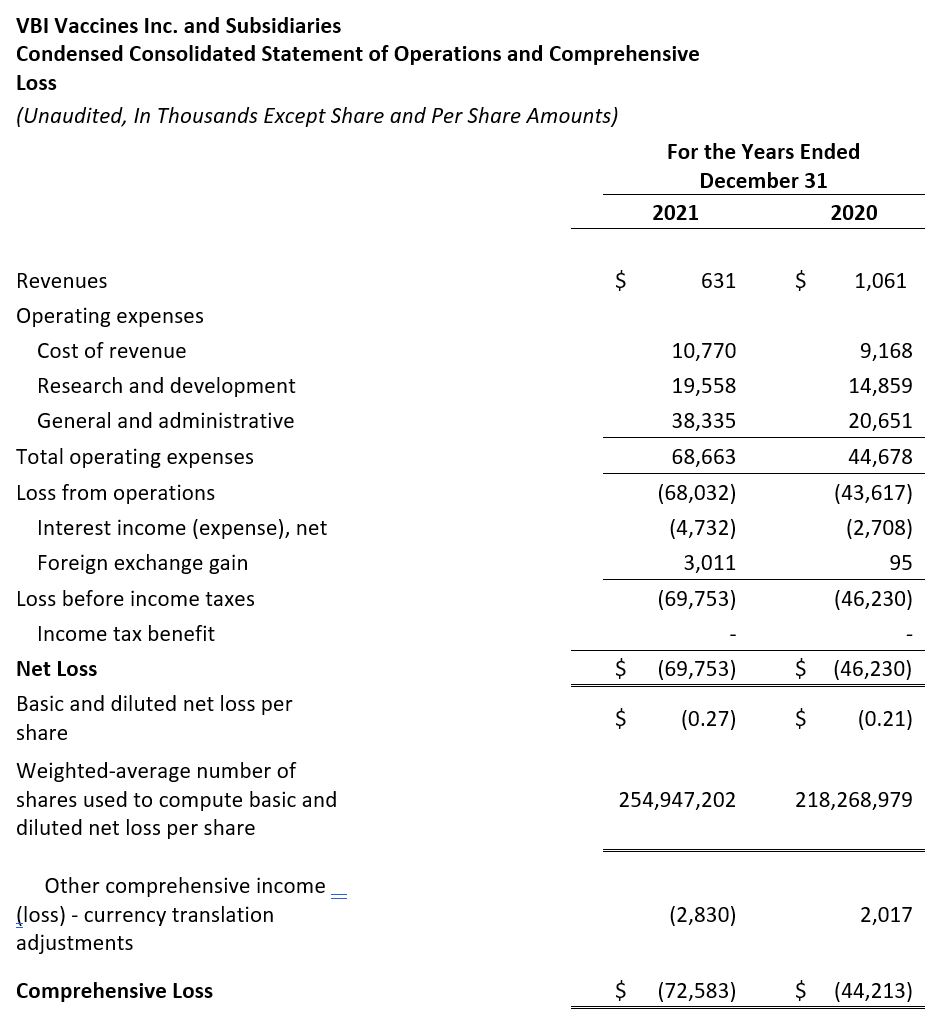
- Revenue: Revenue for the full year 2021 was $0.6 million compared to $1.1 million for the same time period in 2020. The decrease in revenue was due to a decrease in the R&D services revenue earned as part of the License Agreement with Brii Bio in 2021 compared to 2020.
- Research and Development (R&D): R&D expenses for the full year 2021 were $19.6 million compared to $14.9 million for the same period in 2020. The increase in R&D spend was mainly a result of the increase in costs related to the continued development of our eVLP vaccine candidates and increased regulatory costs related to our 3-antigen HBV vaccine.
- General and Administrative (G&A): G&A expenses for full year 2021 were $38.4 million compared to $20.7 million for the same period in 2020. The increase in G&A expense in 2021 was a result of the increase in pre-commercialization activities related to our 3-antigen HBV vaccine, increased insurance costs, and increased professional and labor costs.
- Net Loss: Net loss and net loss per share for the year ended December 31, 2021 were $69.8 million and $0.27, respectively, compared to a net loss of $46.2 million and a net loss per share of $0.21 for the year ended December 31, 2020.
About PreHevbrio™
PreHevbrio is an adult hepatitis B vaccine that contains the three hepatitis B surface antigens of the hepatitis B virus – S, pre-S1, and pre-S2. PreHevbrio is also approved for use and commercially available in Israel under the brand name Sci-B-Vac®.
U.S. Indication and Use
PreHevbrio is indicated for prevention of infection caused by all known subtypes of hepatitis B virus. PreHevbrio is approved for use in adults 18 years of age and older.
U.S. Important Safety Information (ISI)
Do not administer PreHevbrio to individuals with a history of severe allergic reaction (e.g. anaphylaxis) after a previous dose of any hepatitis B vaccine or to any component of PreHevbrio.
Appropriate medical treatment and supervision must be available to manage possible anaphylactic reactions following administration of PreHevbrio.
Immunocompromised persons, including those on immunosuppressant therapy, may have a diminished immune response to PreHevbrio.
PreHevbrio may not prevent hepatitis B infection, which has a long incubation period, in individuals who have an unrecognized hepatitis B infection at the time of vaccine administration.
The most common side effects (> 10%) in adults age 18-44, adults age 45-64, and adults age 65+ were pain and tenderness at the injection site, myalgia, fatigue, and headache.
There is a pregnancy exposure registry that monitors pregnancy outcomes in women who received PreHevbrio during pregnancy. Women who receive PreHevbrio during pregnancy are encouraged to contact 1-888-421-8808 (toll-free).
To report SUSPECTED ADVERSE REACTIONS, contact VBI Vaccines at 1-888-421-8808 (toll-free) or VAERS at 1-800-822-7967 or www.vaers.hhs.gov.
Please see Full Prescribing Information.
References
- Qiu K, Liu B, Li S-Y, et. al. Systematic review with meta-analysis: combination treatment of regimens based on pegylated interferon for chronic hepatitis B focusing on hepatitis B surface antigen clearance. Alimentary Pharmacology & Therapeutics 2018;47:1340-1348.
Condensed Consolidated Financial Statements
About VBI Vaccines Inc.
VBI Vaccines Inc. (“VBI”) is a biopharmaceutical company driven by immunology in the pursuit of powerful prevention and treatment of disease. Through its innovative approach to virus-like particles (“VLPs”), including a proprietary enveloped VLP (“eVLP”) platform technology, VBI develops vaccine candidates that mimic the natural presentation of viruses, designed to elicit the innate power of the human immune system. VBI is committed to targeting and overcoming significant infectious diseases, including hepatitis B, coronaviruses, and cytomegalovirus (CMV), as well as aggressive cancers including glioblastoma (GBM). VBI is headquartered in Cambridge, Massachusetts, with research operations in Ottawa, Canada, and a research and manufacturing site in Rehovot, Israel.
VBI Contact
Nicole Anderson
Director, Corporate Communications & IR
Phone: (617) 830-3031 x124
Email: IR@vbivaccines.com
Cautionary Statement on Forward-looking Information
Certain statements in this press release that are forward-looking and not statements of historical fact are forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and are forward-looking information within the meaning of Canadian securities laws (collectively, “forward-looking statements”). The Company cautions that such statements involve risks and uncertainties that may materially affect the Company’s results of operations. Such forward-looking statements are based on the beliefs of management as well as assumptions made by and information currently available to management. Actual results could differ materially from those contemplated by the forward-looking statements as a result of certain factors, including but not limited to, the impact of general economic, industry or political conditions in the United States or internationally; the impact of the ongoing COVID-19 pandemic on our clinical studies, manufacturing, business plan, and the global economy; the ability to successfully manufacture and commercialize PreHevbrio; the ability to establish that potential products are efficacious or safe in preclinical or clinical trials; the ability to establish or maintain collaborations on the development of pipeline candidates and the commercialization of PreHevbrio; the ability to obtain appropriate or necessary regulatory approvals to market potential products; the ability to obtain future funding for developmental products and working capital and to obtain such funding on commercially reasonable terms; the Company’s ability to manufacture product candidates on a commercial scale or in collaborations with third parties; changes in the size and nature of competitors; the ability to retain key executives and scientists; and the ability to secure and enforce legal rights related to the Company’s products. A discussion of these and other factors, including risks and uncertainties with respect to the Company, is set forth in the Company’s filings with the SEC and the Canadian securities authorities, including its Annual Report on Form 10-K filed with the SEC on March 7, 2022, and filed with the Canadian security authorities at sedar.com on March 7, 2022, as may be supplemented or amended by the Company’s Quarterly Reports on Form 10-Q. Given these risks, uncertainties and factors, you are cautioned not to place undue reliance on such forward-looking statements, which are qualified in their entirety by this cautionary statement. All such forward-looking statements made herein are based on our current expectations and we undertake no duty or obligation to update or revise any forward-looking statements for any reason, except as required by law.









