- Q4 2020: Sci-B-Vac® regulatory approval submissions in the US, Europe, and Canada expected to begin
- Q2 2020 and Q4 2020: Data from Phase 2a of VBI’s cancer vaccine immunotherapeutic, VBI-1901, are expected
- H2 2020: Data from Phase 1b/2a study of hepatitis B immunotherapeutic, VBI-2601 (BRII-179) expected
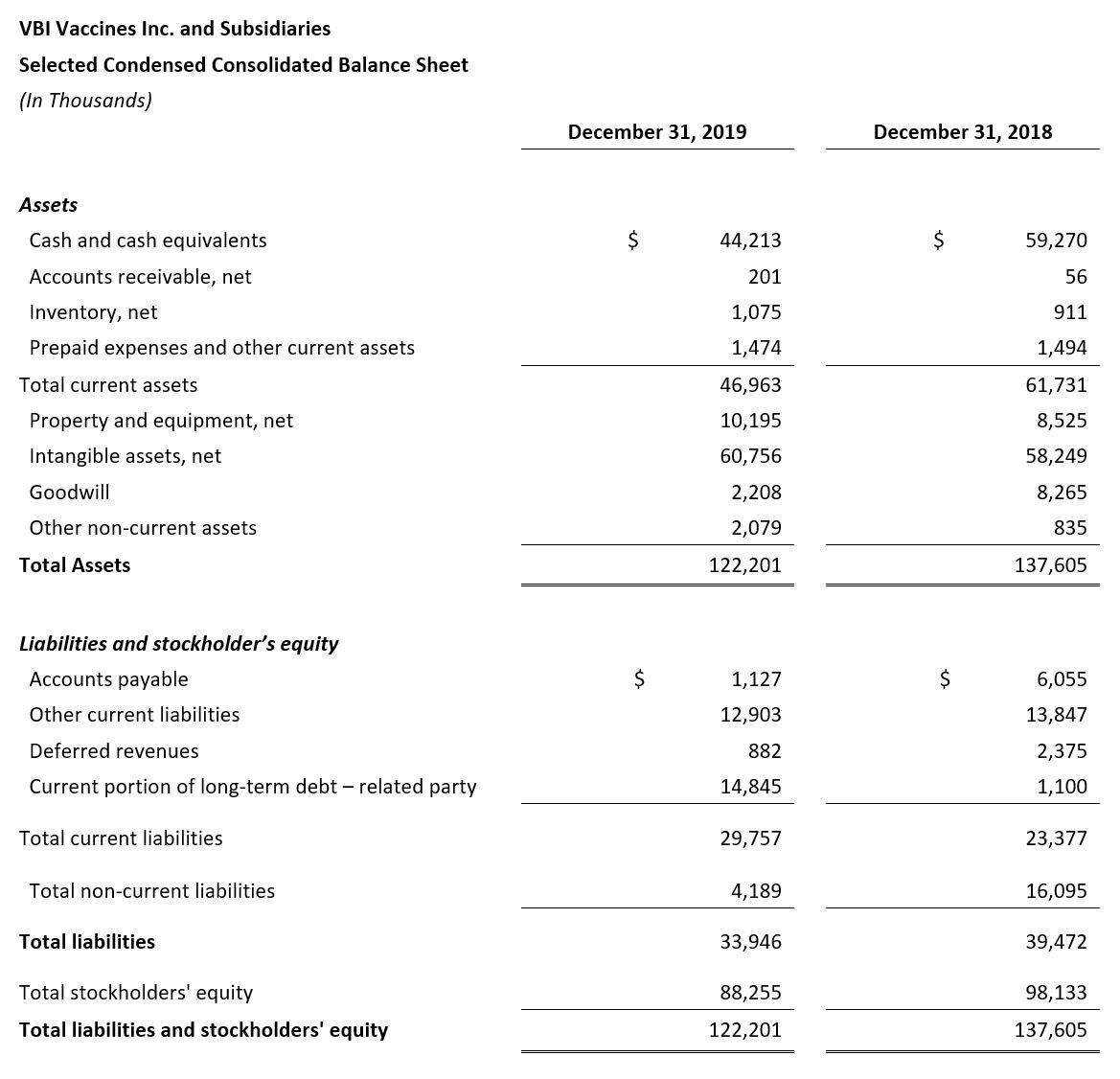
- 2019 year-end cash position of $44.2 million
VBI Vaccines Inc. (NASDAQ: VBIV) (“VBI”), a commercial-stage biopharmaceutical company developing next-generation infectious disease and immuno-oncology vaccines, today provided a corporate update, its outlook for 2020, and announced financial results for the fourth quarter and twelve months ended December 31, 2019.
Annual Note from Jeff Baxter, President and CEO
“Over the last 18 months, VBI has transformed into a company committed to changing the landscape of hepatitis B (HBV) by improving prevention and working to develop a functional cure. HBV is a significant global public health issue with an increasing disease burden – by example, the acute HBV infection rate in the US increased by 20.7% in 2015, rising for the first time since 2006, with the sharpest increases occurring largely in states that have been impacted most by the ongoing opioid epidemic1. More than that, HBV is often an asymptomatic virus – it is estimated that as many as 67% of people with chronic HBV in the US are unaware of their infection status2. All of this underlines the importance of vaccination against HBV.
“Our trivalent hepatitis B vaccine, Sci-B-Vac®, has successfully completed the pivotal Phase 3 program, demonstrating its ability to safely and rapidly elicit robust immune responses, conferring seroprotection in adults, including those who have been harder to protect, namely older adults, diabetics, and obese individuals. We are now working with the FDA and other regulatory agencies to prepare for submissions of regulatory approval applications in the US, Europe, and Canada, beginning in the fourth quarter of 2020. We believe Sci-B-Vac has the potential to be an efficacious, safe, and cost-effective option for all adults seeking protection against HBV.
“While we believe prevention is always better, and more cost-effective, we also recognize the importance of a cure for HBV. Recent estimates of people chronically-infected with HBV worldwide range from 240 million to 350 million, with over 2 million in the US alone3; globally, more than 2 billion people have ever been infected with HBV, acutely or chronically4. The race to develop a functional cure for HBV is competitive, but we believe consensus amongst experts is building that an immunotherapeutic would be needed to achieve long-term immunologic control and restore the body’s defense against HBV. With our new trivalent formulation that enhances T-cell response, we believe VBI-2601 (BRII-179) is well-positioned to be the immunotherapeutic component of a functional cure. Initial data from the ongoing Phase 1b/2a clinical study for VBI-2601, conducted with our partner Brii Biosciences (Brii Bio), are expected in the second half of 2020.
“In our other programs, we are integrating our cytomegalovirus (CMV) expertise with our proprietary enveloped virus-like particle (eVLP) technology to develop therapeutic cancer vaccines and prophylactic vaccines. Our lead eVLP program, VBI-1901, targets the most aggressive form of adult brain cancer, glioblastoma (GBM), a CMV-associated solid tumor. GBM is a devastating disease leaving patients with few treatments options. Encouraging data from the ongoing Phase 1/2a study in recurrent GBM patients were presented throughout 2019, with demonstrated correlations between immunologic responses, tumor responses (assessed through MRI scans), and clinical responses (survival data). As a testament to the strength of the early data, we entered into a collaboration with GlaxoSmithKline (GSK) to clinically evaluate the combination of VBI-1901 with GSK’s proprietary AS01B adjuvant system, a highly-innovative adjuvant system that has contributed to positive results in GSK’s shingles vaccine, Shingrix. We expect additional data from Part B of the ongoing Phase 1/2a study throughout 2020. We hope to continue to see encouraging results from VBI-1901 as we work hard to provide meaningful benefit to these patients.
“There were numerous clinical milestones throughout 2019, but we believe 2020 will be an even more transformational year for the company. The VBI team in the US, Canada, and Israel are working hard to address significant unmet medical needs, and we believe this dedication and focus on successfully achieving fundamentals will drive shareholder value. We appreciate the continued shareholder support as well as that of our partners and volunteers who participate in our clinical trials, and we look forward to continued advancement and achievement in 2020 and beyond.”
Recent Key Program Achievements and Projected Upcoming Milestones
Sci-B-Vac®: Trivalent Prophylactic HBV Vaccine
- June 2019 and January 2020 : Announcement of positive top-line results from both pivotal Phase 3 studies which, combined, showed:
- Non-inferiority of seroprotection rate (SPR) in all adults ≥ age 18 compared with Engerix-B®;
- Superiority of SPR in adults ≥ age 45 compared with Engerix-B®;
- Higher SPR in all key subgroup analyses of adults ≥ 18 years, including by age, gender, body mass index, diabetic status, and smoking status, compared with Engerix-B®;
- Higher SPR at all time points compared to Engerix-B® and, in CONSTANT, non-inferiority of SPR after 2 doses of Sci-B-Vac® compared with 3 doses of Engerix-B® in adults age 18-45 years;
- Lot-to-lot manufacturing consistency; and
- Safety and tolerability consistent with the known safety profile of Sci-B-Vac®, with no new safety risks identifies and no safety signals observed.
- Q2 2020 : Pre-BLA discussions expected with the FDA.
- Q4 2020 : Submission of applications for regulatory approvals in the US, Europe, and Canada expected to begin.
VBI-2601 (BRII-179): HBV Immunotherapeutic Candidate
- November 2019 : Initiation of enrollment in Phase 1b/2a study in patients with chronic HBV infection. The Phase 1b/2a is a randomized, controlled study designed to assess the safety, tolerability, antiviral, and immunological activity of VBI-1901 (BRII-179). The study is expected to enroll up to 65 patients and is sponsored by our partner Brii Bio.
- H2 2020 : Initial human proof-of-concept data expected from Phase 1b/2a study.
VBI-1901: Cancer Vaccine Immunotherapeutic Candidate
- June 2019, November 2019, and March 2020: Encouraging data from the ongoing Phase 1/2a clinical study of VBI-1901 in recurrent GBM patients were announced, demonstrating that:
- VBI-1901 is well-tolerated at all doses tested, with no vaccine-related safety signals observed;
- Two patients in the high-dose cohort of Part A experienced a 60% reduction in the size of the primary tumor;
- In the high-dose cohort of Part A, vaccine response correlated with clinical response, with a 12-month overall survival (OS) rate of 83% (n=5/6) for vaccine responders vs. 33% (n=3/9) for non-responders – vaccine responders also saw a 6.25-month improvement in median OS (14.0 months) compared with vaccine non-responders (7.75 months); and
- Early tumor and immunologic responses in Part B aligned with responses and benefit observed in Part A.
- September 2019 : Announcement of collaboration with GSK to clinically evaluate VBI-1901 in combination with AS01B as a second arm in Part B of the ongoing Phase 1/2a study in first-recurrent GBM patients
- February 2020 : Enrollment initiated in VBI-1901 + GSK’s AS01B study arm
- Q2 2020 : Expanded immunologic, tumor, and clinical data expected from Phase 2a VBI-1901 + GM-CSF study arm
- Q4 2020 : Initial immunologic and tumor response data expected from Phase 2a VBI-1901 + AS01B study arm
Financial Results for the Three and Twelve Months Ended December 2019
- Cash Position: VBI ended the fourth quarter of 2019 with $44.2 million cash and cash equivalents compared with $59.3 million as of December 31, 2018.
- Net Cash Used in Operating Activities: Net cash used in operating activities for the full year 2019 was $48.7 million, compared to $45.5 million for the same period in 2018.
- Cash Used for Purchase of Property and Equipment: The purchase of property and equipment in 2019 was $3.7 million, compared to $6.0 million in 2018, and was primarily in Rehovot, Israel, as part of the modernization and capacity increase of the manufacturing facility. As part of this process, manufacturing at the facility in Rehovot, Israel, was temporarily shut-down in 2018 and re-commenced in May 2019. Following completion of the modernization and capacity increase, VBI received the certificate of Good Manufacturing Practice (GMP) compliance from the Israeli Ministry of Health in January 2020.
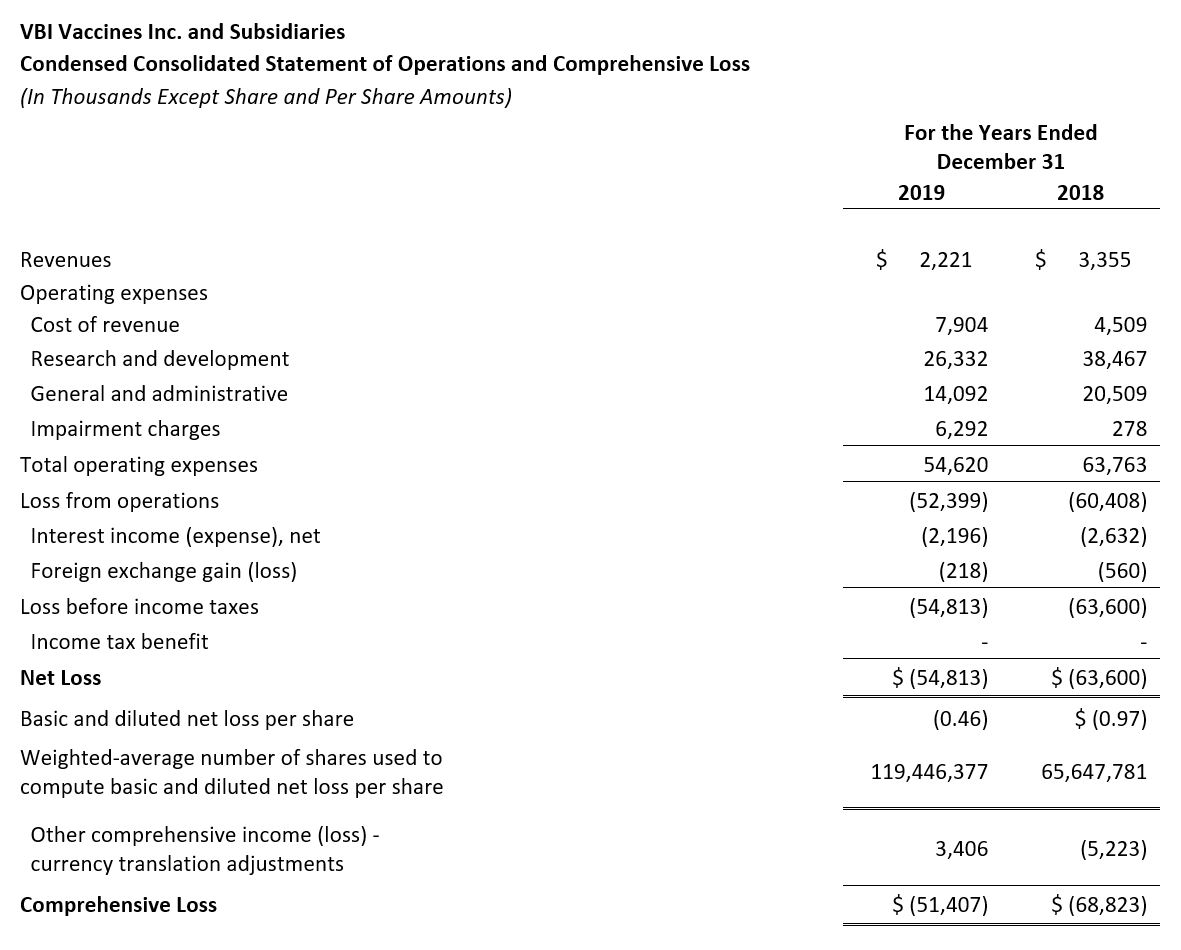
- Revenue: Revenue for the three months ended December 31, 2019 and for the full year 2019 was $0.6 million and $2.2 million, respectively, compared to $2.7 million and $3.4 million for the same time periods in 2018, respectively. The decrease in revenue was due to decreased named-patient product sales in Europe in 2019. Additionally, there was a decrease as a result of the license revenue earned as part of the License Agreement with Brii Bio in 2018, but this was offset by an increase in R&D services revenue in 2019 as part of the same agreement with Brii Bio.
- Research and Development (R&D): R&D expenses for the fourth quarter and full year 2019 were $4.3 million and $26.3 million, respectively, compared to $10.1 million and $38.5 million for the same periods in 2018, respectively. The decrease in R&D spend in 2019 was primarily due to a decrease in costs as the Phase 3 program for Sci-B-Vac® neared completion.
- General and Administrative (G&A): G&A expenses for the fourth quarter and full year 2019 were $3.8 million and $14.1 million, respectively, compared to $9.9 million and $20.5 million for the same periods in 2018, respectively. The decrease in G&A expense in 2019 was primarily due to a $6 million payment made in 2018 to re-obtain Sci-B-Vac® distribution rights in Asia, with no similar payment made during 2019. Other variances include decreased administrative expenses and the re-allocation of expenses from G&A back to Cost of Goods in 2019 related to the temporary Rehovot facility closure.
- Impairment Charge: There was an impairment charge of $6.3 million in 2019 related to goodwill, compared to $0.3 million in 2018.
- Net Loss: Net Loss and net loss per share for the year ended December 31, 2019 were $54.8 million and $0.46, respectively, compared to a net loss of $63.6 million and a net loss per share of $0.97 for the year ended December 31, 2018.
- Pacho R. The Rise in Acute Hepatitis B Infection in the U.S.; U.S. Department of Health & Human Services, HHS.gov; February 21, 2018
- Kim HS, Yang JD, El-Serag HB, Kanwal F. Awareness of chronic viral hepatitis in the United Stated: An update from the National Health Nutrition Examination Survey. J Viral Hepat. 2019 May; 26(5):596-602
- Schillie S, Vellozzi C, Reingold A, et al. Prevention of Hepatitis B Virus Infection in the United States: Recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep 2018;67(No. RR-1):1-31. DOI: http://dx.doi.org/10.15585/mmwr.rr6701a1
- MacLachlan JH, Cowie BC. Hepatitis B Virus Epidemiology. Cold Sprin Harb Perspect Med. 2015 May; 5(5): a021410
About VBI Vaccines Inc.
VBI Vaccines Inc. (Nasdaq: VBIV) is a commercial-stage biopharmaceutical company developing next generation vaccines to address unmet needs in infectious disease and immuno-oncology. VBI is advancing the prevention and treatment of hepatitis B, with the only commercially-approved trivalent hepatitis B vaccine, Sci-B-Vac®, which is approved for use in Israel, and recently completed its Phase 3 program in the U.S., Europe, and Canada. VBI is also developing an immunotherapeutic candidate as part of a functional cure for chronic hepatitis B. VBI’s enveloped virus-like particle (eVLP) Platform technology enables development of eVLPs that closely mimic the target virus to elicit a potent immune response. VBI’s lead eVLP program candidates include a vaccine immunotherapeutic candidate targeting glioblastoma (GBM), a cytomegalovirus (CMV)-associated tumor, and a prophylactic CMV vaccine candidate. VBI is headquartered in Cambridge, MA with research operations in Ottawa, Canada and research and manufacturing facilities in Rehovot, Israel.
Cautionary Statement on Forward-looking Information
Certain statements in this press release that are forward-looking and not statements of historical fact are forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and are forward-looking information within the meaning of Canadian securities laws (collectively, “forward-looking statements”). The company cautions that such statements involve risks and uncertainties that may materially affect the company’s results of operations. Such forward-looking statements are based on the beliefs of management as well as assumptions made by and information currently available to management. Actual results could differ materially from those contemplated by the forward-looking statements as a result of certain factors, including but not limited to the ability to establish that potential products are efficacious or safe in preclinical or clinical trials; the ability to establish or maintain collaborations on the development of therapeutic candidates; the ability to obtain appropriate or necessary governmental approvals to market potential products; the ability to successfully commercialize our product candidates following regulatory approvals; the ability to obtain future funding for developmental products and working capital and to obtain such funding on commercially reasonable terms; the company’s ability to manufacture product candidates on a commercial scale or in collaborations with third parties; changes in the size and nature of competitors; the ability to retain key executives and scientists; and the ability to secure and enforce legal rights related to the company’s products. A discussion of these and other factors, including risks and uncertainties with respect to the company, is set forth in the Company’s filings with the Securities and Exchange Commission and the Canadian securities authorities, including its Annual Report on Form 10-K filed with the Securities and Exchange Commission and with the Canadian security authorities at sedar.com, as may be supplemented or amended by the Company’s Quarterly Reports on Form 10-Q. Given these risks, uncertainties and factors, you are cautioned not to place undue reliance on such forward-looking statements, which are qualified in their entirety by this cautionary statement. All such forward-looking statements made herein are based on our current expectations and we undertake no duty or obligation to update or revise any forward-looking statements for any reason, except as required by law.
VBI Contact
Nicole Anderson
Director, Corporate Communications & IR
Phone: (617) 830-3031 x124
Email: IR@vbivaccines.com









